 |
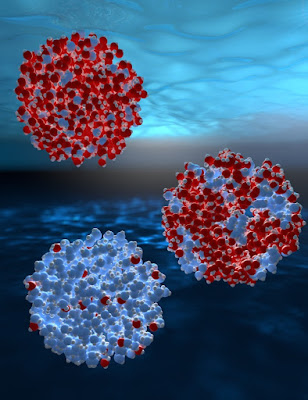
| Illustration showing fluctuations between regions of two different local structures of water. High density is shown as red and low density as blue. (Courtesy: Stockholm University) |
Topics: Fluid Mechanics, Materials Science, Thermodynamics
Water could exist in two different liquid phases with different densities. That is the conclusion of researchers in Sweden, Japan and Korea, who have used ultrafast X-ray scattering to measure the properties of supercooled water droplets.
Despite being the most ubiquitous and important liquid on Earth, water is a deeply puzzling substance with physical properties that deviate significantly from those of an idealized liquid. Several theories have been advanced to account for some of water’s idiosyncrasies, but experimental data have been lacking.
Solid ice is the most stable phase of water below 0° C, but the liquid phase remains metastable at sub-zero temperatures. Under normal circumstances, impurities such as dust particles provide nuclei around which ice crystals can form, so freezing occurs quickly. In the laboratory, however, it is relatively easy to supercool liquid water to well below 0° C by removing impurities. As the temperature goes down further, however, molecular motion slows and, below around -40° C, water molecules begin to form crystals around one another, allowing even pure water to crystallize very rapidly.
Supercooled water could exist in two liquid phases, Tim Wogan, Physics World
Comments