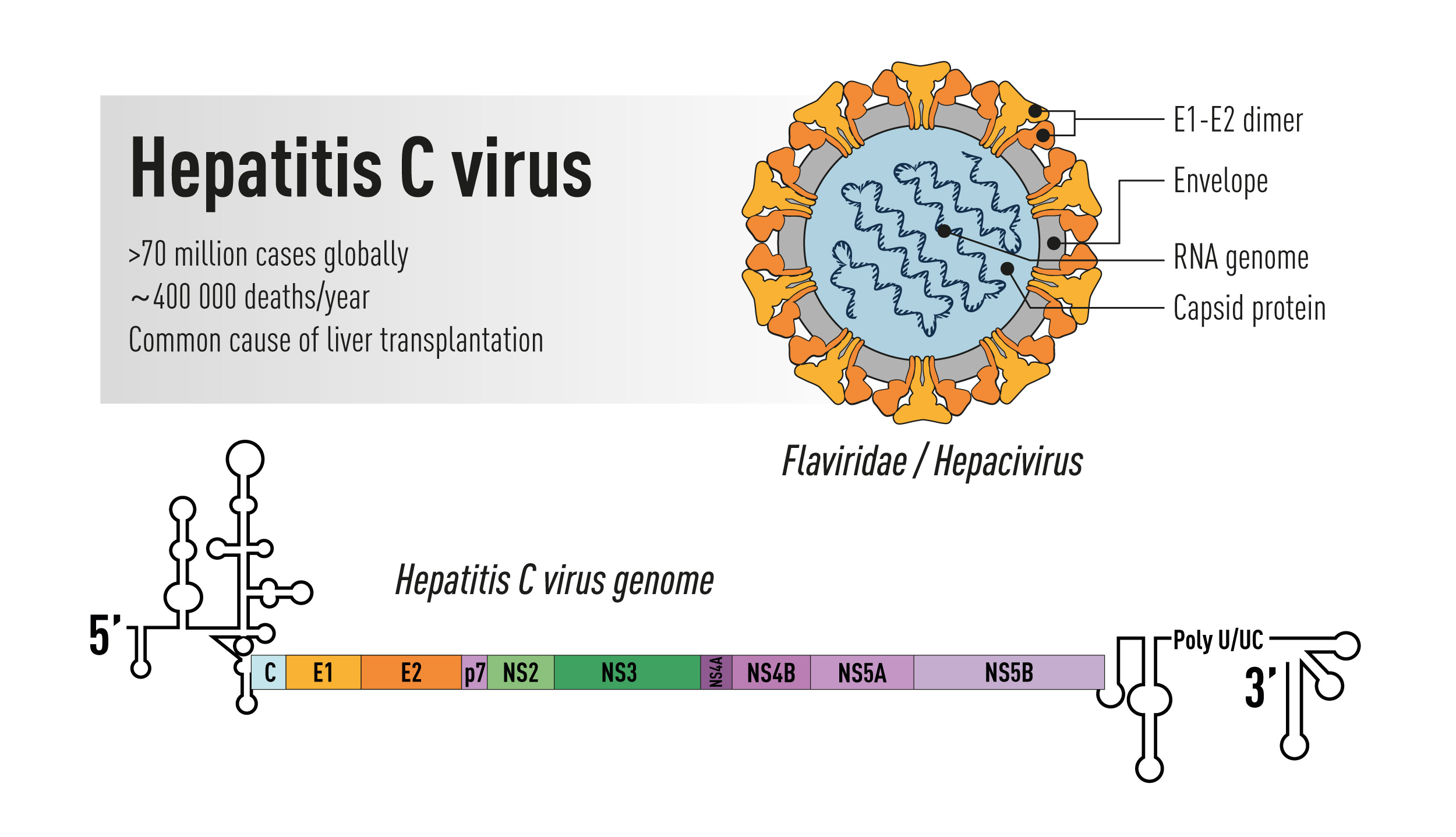
Topics: African Americans, Biology, Black History Month, Civics, Civil Rights, Civilization, Diversity in Science, Medicine
“Father of the Blood Bank”
June 3, 1904 – April 1, 1950
Renowned surgeon and pioneer in the preservation of life-saving blood plasma
Major scientific achievements:
- Discovered method for long-term storage of blood plasma
- Organized America's first large-scale blood bank
Dr. Charles Richard Drew broke barriers in a racially divided America to become one of the most important scientists of the 20th century. His pioneering research and systematic developments in the use and preservation of blood plasma during World War II not only saved thousands of lives but innovated the nation’s blood banking process and standardized procedures for long-term blood preservation and storage techniques adapted by the American Red Cross.
A native Washingtonian, Drew was an average student but a gifted athlete recruited in 1922 on a football and track and field scholarship by Amherst College in Massachusetts. He was one of only 13 African Americans in a student body of 600, where the racial climate exposed him to hostility from opposing teams. His own football team passed him over as captain his senior year even though he was the team’s best athlete.
Beyond sports, Drew didn’t have a clear direction until a biology professor piqued his interest in medicine. Like many other fields, medicine was largely segregated, greatly limiting education and career options for African Americans. For Drew, the narrowed road would lead him to McGill University College of Medicine in Montréal. There, he distinguished himself, winning the annual scholarship prize in neuroanatomy; becoming elected to the medical honor society Alpha Omega Alpha; and staffing the McGill Medical Journal. He also won the J. Francis Williams Prize in medicine after beating the top 5 students in an exam competition. In 1933, Drew received his MD and CM (Master of Surgery) degrees, graduating second in a class of 137.
Drew’s interest in transfusion medicine began during his internship and surgical residency at Montreal Hospital (1933-1935) working with bacteriology professor John Beattie on ways to treat shock with fluid replacement. Drew aspired to continue training in transfusion therapy at the Mayo Clinic, but racial prejudices at major American medical centers barred black scholars from their practices. He would instead join the faculty at Howard University College of Medicine, starting as a pathology instructor, and then progressing to surgical instructor and chief surgical resident at Freedmen's Hospital.
Dr. Charles Richard Drew, American Chemical Society